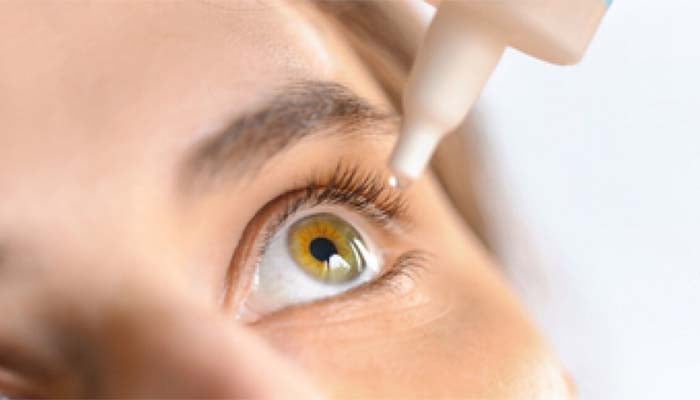
The K.C Pharmaceutical has voluntarily recalled more than three million bottles of eye drops distributed nationwide.
The US Food and Drug Administration (FDA) reported that the California pharmaceutical company has recalled 3.1 millions eye drop products, citing “lack of assurance of sterility” in items.
The recalled eye drops were distributed by companies, including Kroger, Walgreens, H-E-B, Military Exchanges, and more.
Notably, the FDA has classified the recall as “Class II,” meaning the use of this product is likely to pose reversible health issues.
The affected lot of recalled eyedrops include:
- 182,000 bottles of Sterile Eye Drops AC
- 303,000 bottles of Eye Drops Advanced Relief
- 1,024,000 bottles of Dry Eye Relief Eye Drops
- 245,000 bottles of Ultra Lubricating Eye Drops
- 378,000 bottles of Sterile Eye Drops Original Formula
- 315,000 bottles of Sterile Eye Drops Redness Lubricant
- 74,000 bottles of Sterile Eye Drops Soothing Tears
- 590,000 bottles of Artificial Tears Sterile Lubricant Eye Drops
Earlier this year, a recall of eye drops was filed due to “presence of glass-like particles” in the products.
If anyone has already used these drops and experiences eye pain, redness, or vision changes are advised to contact an ophthalmologist or healthcare provider immediately.