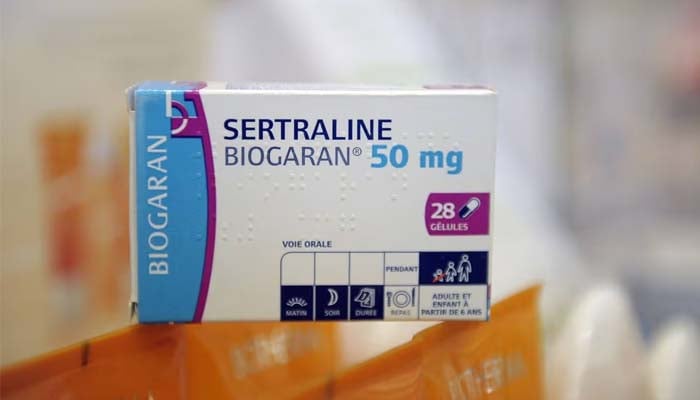
Over 8,000 packs of a frequently used antidepressant have been recalled after a patient found the wrong medicine inside their pack.
The recall comes after a complaint by a patient who consumed Sertraline 100 mg film-coated tablets and rather discovered a strip of another antidepressant, Citalopram 40mg film-coated tablets, inside the sealed box.
Both the medicines are selective serotonin reuptake inhibitors (SSRIs) used for the treatment of anxiety disorders, depression, and related mental health disorders by boosting serotonin to the brain.
The UK-based pharmaceutical company, Amarox, has issued a "precautionary recall" of a particular batch of Sertraline, according to the Medicines and Healthcare products Regulatory Agency (MHRA).

The same manufacturer produces both medications at the same site.
The MHRA said that the manufacturing error “appears to have occurred during secondary packaging of the blister strips into the cartons”.
They said that patients who have already taken any Citalopram 40mg tablets by mistake or are experiencing side effects should seek medical advice immediately.
The MHRA said that pharmacists should stop supplying the batch and contact any patients who may have been dispensed the impacted product, Sertraline 100mg tablets batch number V2500425.
The regulator said 81,872 packs are included in the recall.
The affected lot numbers of the recalled tablets include V2500425.
Experts urged to check batch numbers and expiration dates before buying and consuming the tablets.
In 2019, over 16.7 million prescriptions of Sertraline were issued by GPs in England, as per research.